
Thus, water is H2O and carbon dioxide is CO2. By convention, no subscript is written when a molecule contains only one atom of an element. The shorthand notation for a compound describes the number of atoms of each element, which is indicated by a subscript written after the symbol for the element. Nonmetals combine with each other to form covalent compounds, which exist as neutral molecules. These compounds are composed of positive and negative ions formed by adding or subtracting electrons from neutral atoms and molecules. Metals often react with nonmetals to form ionic compounds. When a compound decomposes, the atoms are recovered unchanged Compounds Elements combine to form chemical compounds that are often divided into two categories. Atoms of different elements combine in simple whole numbers to form compounds. A compound can be decomposed by chemical. Atoms of different elements have different weights and different chemical properties. A pure substance that contains two or more elements chemically combined together in a fixed ratio. 
Matter is made up of atoms that are indivisible and indestructible. 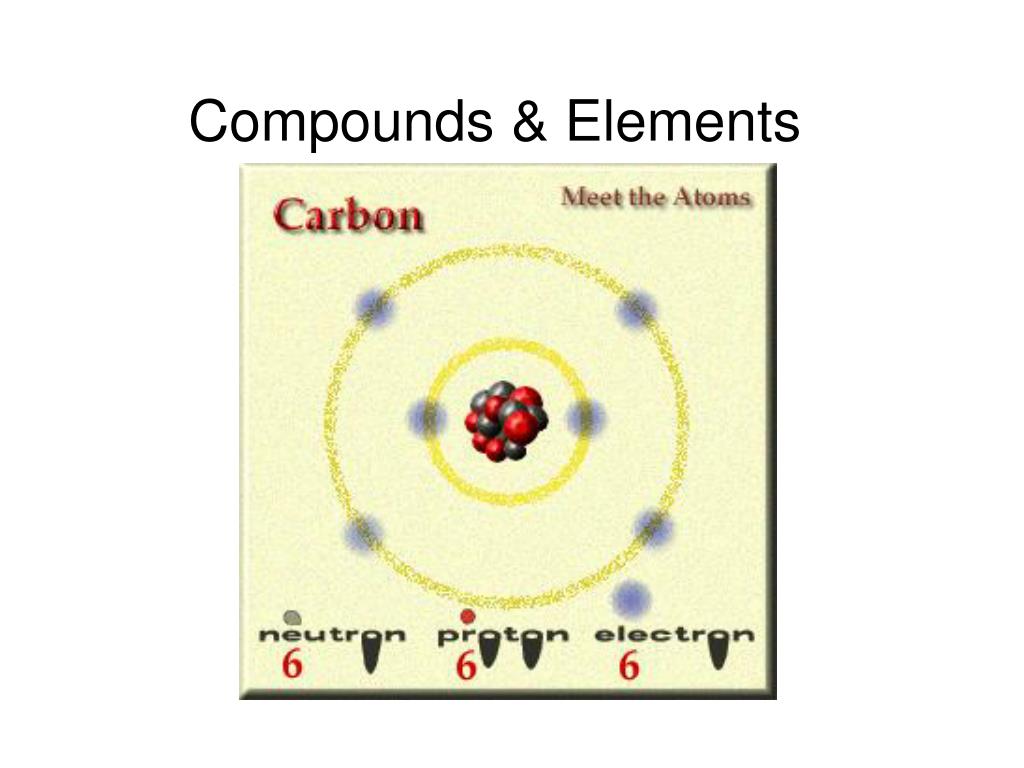
Atoms Elements are made up of atoms, the smallest particle that has any of the properties of the element.John Dalton, in 1803, proposed a modern theory of the atom based on the following assumptions. The meaning of COMPOUND is something formed by a union of elements or parts especially : a distinct substance formed by chemical union of two or more. The semimetals can be found along the dividing line between the metals and the nonmetals. A handful of nonmetals are clustered in the upper right corner of the periodic table. Most elements are metals, which are found on the left and toward the bottom of the periodic table. The elements can be divided into three categories that have characteristic properties: metals, nonmetals, and semimetals. Any readily measurable mass of an element or compound contains an extraordinarily large number of atoms, molecules, or ions, so an extraordinarily large numerical unit is needed to count them. The notation for each element can be found on the periodic table of elements. Each element is represented by a unique symbol. They are therefore the elementary, or simplest, chemical substances - elements. Hydrogen and oxygen, on the other hand, cannot be decomposed into simpler substances. Example: Water decomposes into a mixture of hydrogen and oxygen when an electric current is passed through the liquid. Because atoms cannot be created or destroyed in a chemical reaction, elements such as phosphorus (P4) or sulfur (S8) cannot be broken down into simpler substances by these reactions. The difference between an element and a compound is that an element is a substance made of same type of atoms, whereas a compound is made of different elements. Any substance that contains only one kind of an atom is known as an element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
